In the News
IPA Releases New HYFT-Powered In Silico Humanization Platform, Aims to Disrupt the Transgenic Animal Model Market
VICTORIA, British Columbia, June 6, 2023 – ImmunoPrecise Antibodies Ltd. (NASDAQ: IPA) (“IPA” or the “Company”) today introduced a transformative AI-driven rapid therapeutic screening platform, the result of a collaboration between IPA Canada and its subsidiary, BioStrand®. This innovative solution aims to significantly expedite the early stages of drug discovery by enabling the early elimination of less promising therapeutic candidates, thereby reducing time, cost, and the risk of failure during later stage discovery.
The current transgenic animal model market, reaching USD 17.7 billion in 2021 and expected to grow by 8.1% CAGR from 2022 to 2028¹, has been widely adopted in novel therapeutic discovery, primarily to avoid what was previously seen as time-consuming, expensive, risk-laden, and sometimes unsuccessful humanization processes. However, despite its widespread adoption, transgenics pose limitations of their own, such as a lack of diversity in therapeutic outputs, loss of therapeutic efficacy, and high costs. Addressing these challenges, BioStrand's recently patented technology introduces a reliable, scalable, and cost-effective alternative for early-stage therapeutic discovery.
BioStrand’s platform, termed the HYFT® Proprietary Proteome Presence Assessment, optimizes potential therapeutic drug candidates at an early stage using the multi-functional LENSai™ in silico humanization platform integrated with its patented HYFT technology. This allows a multitude of potential therapies to be analyzed concurrently, reducing analysis time and costs, and ensuring that only the most promising candidates proceed further into the drug discovery process.
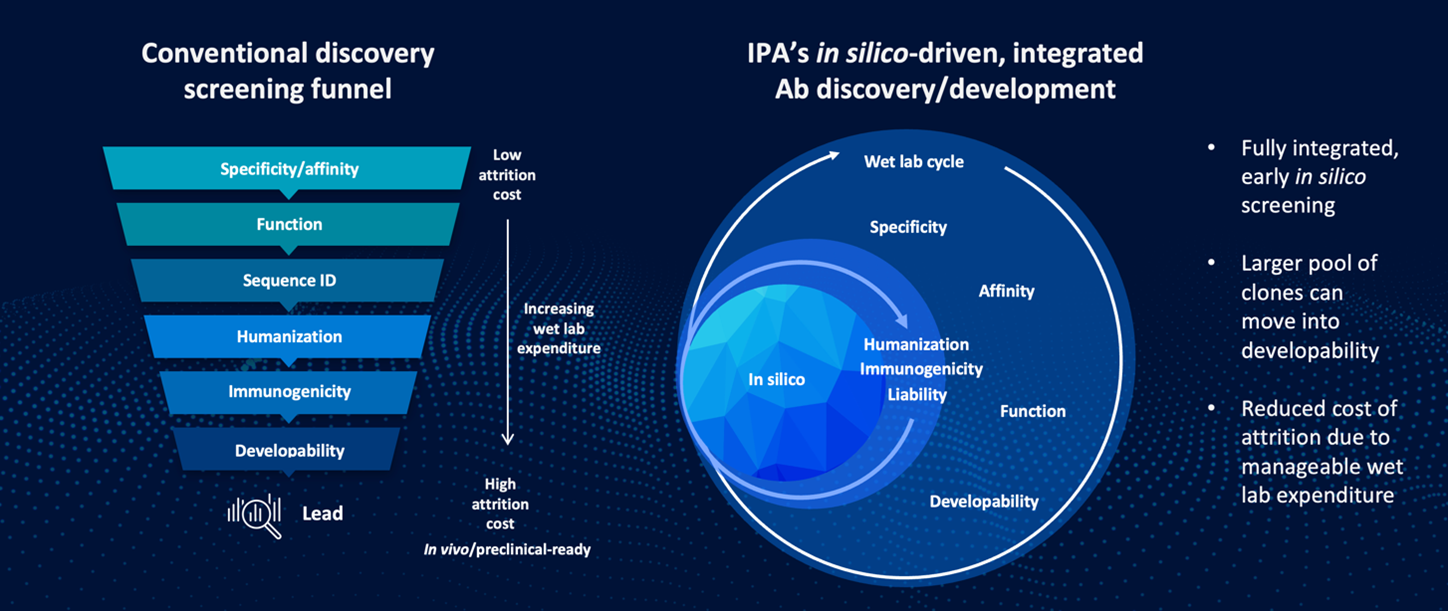
(LEFT) Traditional antibody discovery and development timeline. Linear model imposes increased risk of attrition due to the cost of lead candidate advancement with minimal insight into future clinical viability.
(RIGHT) The new enhanced predictive insights gained by IPA’s new in silico-driven discovery cycle, which increases the probability of clinical success, saving our partners time and money.
The concept of in silico humanization refers to the computational process of making an antibody or protein from a non-human source resemble human proteins. This is crucial for reducing the likelihood of immune responses against therapeutics, while preserving their functionality.
BioStrand's patented HYFT technology, incorporated into their proprietary LENSai software, screens the entire human proteome (the entire complement of proteins that is or can be expressed by a cell, tissue, or organism), as well as various animal proteomes, against candidate therapeutics. This screening is accomplished in under one minute per candidate molecule. This process advances the most 'human-like' molecules, speeding up humanization, reducing the manipulation required before use in clinical trials, and offering a rapid, scalable, and cost-effective alternative to transgenic animal models.
Other existing methods can only assess candidates against a subset of the human proteome, which can result in an incomplete evaluation of a protein's humanness and immunogenicity. HYFT technology, however, uniquely addresses this limitation with several distinct advantages, including:
- Use of the most comprehensive human proteome reference set, enabling more thorough comparisons.
- The ability to extract unique Universal Fingerprint™ patterns from biological sequences.
- The capacity to compare the humanized therapeutics to the human proteome, evaluating its similarity to endogenous proteins.
- The ability to capture both sequence and structural information, enabling a better understanding of the protein's function.
- Scalability and high-throughput capabilities, allowing for rapid assessments of numerous candidate proteins against extensive reference datasets.
Leveraging these unique capabilities, the HYFT Proprietary Proteome Presence Assessment offers a more accurate and complete assessment of candidates' humanness and immunogenicity. This supports the development of safer, more effective therapeutic proteins and antibodies while greatly streamlining the drug development process.
"IPA's smooth fusion of computer-driven humanization and immunogenicity testing paves the way for faster development and boosts the potential of new treatment methods," stated Dr. Ingrid Brands, Co-Founder and General Manager of BioStrand. "Our dedication to innovation and pushing the limits of AI-driven therapeutic discovery is represented in this collaboration and integration. We're all about setting new standards in medicine and driving value for our investors.”
IPA has provided insights into its strategic approach for monetizing this breakthrough technology. The plan involves targeting their extensive existing client base, which includes 19 of the top 20 pharmaceutical companies and hundreds of biotechnology firms. These clients typically face substantial costs, when using transgenic animals, to progress a single therapeutic candidate through the commercialization process. These costs often reach tens to hundreds of millions of dollars per molecule, encompassing upfront payments, milestone payments, and commercial royalties. By introducing a more reliable, efficient, and cost-effective solution via IPA's AI-driven platform, the Company is well-positioned to provide greater results and value for its clients, reducing their costs and accelerating their drug discovery timelines. This increase in quality, productivity, and value is expected to translate directly into increased revenue streams for IPA, fostering substantial financial growth for the Company and its investors.
¹ https://www.gminsights.com/industry-analysis/animal-model-market
About BioStrand’s HYFT Technology
BioStrand's HYFT technology extracts unique patterns, known as Universal Fingerprint™ patterns, from the entire biosphere, and integrates them with various data sources, such as scientific papers and medical records. The resulting Knowledge Graph encompasses over 660 million HYFTs and more than 25 billion relations, providing a powerful resource for researchers and AI-driven analysis. The LENSai platform, powered by HYFT technology, takes advantage of the latest advancements in large language models (LLMs) to bridge the gap between syntax (sequences) and semantics (functions). This enables the platform to extract valuable insights from vast amounts of data, without the limitations of traditional LLMs. IPA's support of BioStrand's pioneering work reinforces its commitment to investing in cutting-edge biotechnology solutions with the potential to transform the industry.
About ImmunoPrecise Antibodies Ltd
ImmunoPrecise Antibodies Ltd. has several subsidiaries in North America and Europe including entities such as Talem Therapeutics LLC, BioStrand BV, ImmunoPrecise Antibodies (Canada) Ltd., and ImmunoPrecise Antibodies (Europe) B.V. (collectively, the “IPA Family”). The IPA Family is a biotherapeutic research and technology group that leverages systems biology, multi-omics modeling, and complex artificial intelligence systems to support its proprietary technologies in bioplatform-based antibody discovery. Services include highly specialized, full-continuum therapeutic biologics discovery, development, and out-licensing to support its business partners in their quest to discover and develop novel biologics against the most challenging targets. For further information, visit www.ipatherapeutics.com.
Forward Looking Information
This news release contains forward-looking statements within the meaning of applicable United States securities laws and Canadian securities laws. Forward-looking statements are often identified by the use of words such as “potential”, “plans”, “expects” or “does not expect”, “is expected”, “estimates”, “intends”, “anticipates” or “does not anticipate”, or “believes”, or variations of such words and phrases or state that certain actions, events or results “may”, “could”, “would”, “might” or “will” be taken, occur or be achieved. Forward-looking information contained in this news release includes, but is not limited to, statements relating to the expected outcome on the drug development process of the integration of IPA’s LENSai in silico humanization platform with its HYFT technology, and statements relating to IPA’s expected increased revenue streams and financial growth . In respect of the forward-looking information contained herein, IPA has provided such statements and information in reliance on certain assumptions that management believed to be reasonable at the time.
Forward-looking information involves known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements stated herein to be materially different from any future results, performance or achievements expressed or implied by the forward-looking information. Actual results could differ materially from those currently anticipated due to a number of factors and risks, including, without limitation, the risk that the integration of IPA’s LENSai in silico humanization platform with its HYFT technology may not have the expected results, as well as those risks discussed in the Company’s Annual Information Form dated July 28, 2022 (which may be viewed on the Company’s profile at www.sedar.com), and the Company’s Form 40-F, dated July 29, 2022 (which may be viewed on the Company’s profile at www.sec.gov). Should one or more of these risks or uncertainties materialize, or should assumptions underlying the forward-looking statements prove incorrect, actual results, performance, or achievements may vary materially from those expressed or implied by the forward-looking statements contained in this news release. Accordingly, readers should not place undue reliance on forward-looking information contained in this news release. The forward-looking statements contained in this news release are made as of the date of this release and, accordingly, are subject to change after such date. The Company does not assume any obligation to update or revise any forward-looking statements, whether written or oral, that may be made from time to time by us or on our behalf, except as required by applicable law.
SOURCE ImmunoPrecise Antibodies
Register for IPA Updates
By clicking submit below, you agree that IPA may store and process your personal information to send you email updates. You can find further information in our Privacy Policy.